CC BY-NC-ND 3.0
Not content with the suffering that gum disease and dental caries cause, creationism's divine malevolence used at least one of the pathogens behind those problems to increase suffering a little more by aggrevating rhuematoid arthritis.
That's the inevitable conclusion honest creationist, who rejects the notion of evolution in which pathogenic parasites evolve naturally without intent, malevolent, benign or indifferent, should be drawing from the evidence from a Tokyo Medical and Dental University team of researchers led by Tokuju Okano and Toshihiko Suzuki of the Department of Bacterial Pathogenesis recently published in the International Journal of Oral Science.
This is not the first instance of creationism's divine malevolence multi-tasking its pathogens: in 2017 a team of researchers from The Chinese University of Hong Kong found a link between five oral bacteria, Peptostreptococcus stomatis, Streptococcus anginosus, Parvimonas micra, Slackia exigua and Dialister pneumosintes and the incidence of stomach and eosphageal cancer.
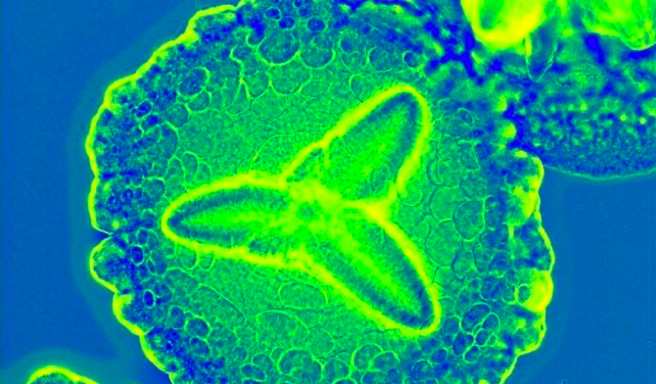
The oral bacterium which has been found to be aggrevating rheumatoid arthritis is Aggregatibacter actinomycetemcomitans.
What information do you have on the oral bacterium Aggregatibacter actinomycetemcomitans? Aggregatibacter actinomycetemcomitans (A. actinomycetemcomitans) is a Gram-negative, facultative anaerobic bacterium commonly found in the human oral cavity. It is a member of the family Pasteurellaceae and is notable for its association with various periodontal diseases, particularly aggressive periodontitis. Key Characteristics:
- Morphology and Structure:
- A. actinomycetemcomitans appears as small, round, non-motile rods.
- It forms characteristic star-shaped colonies on selective culture media.
- The bacterium possesses a distinctive outer membrane with lipopolysaccharides (LPS), which contribute to its virulence and ability to evade the host immune system.
- Virulence Factors:
- Leukotoxin (LtxA): A major virulence factor that can kill neutrophils and other immune cells, helping the bacterium to evade the host immune response.
- Cytolethal distending toxin (CDT): Impairs host cell function and promotes tissue destruction.
- Adhesins: Facilitate adhesion to oral tissues, particularly periodontal tissues.
- Fimbriae: Promote biofilm formation, enhancing bacterial colonization and persistence in the oral cavity.
- Iron acquisition systems: Enable the bacterium to thrive in the iron-limited environment of the human body.
- Pathogenic Role:
- A. actinomycetemcomitans is primarily associated with periodontal diseases, especially localized aggressive periodontitis (LAP), which typically affects younger individuals.
- It contributes to tissue destruction through the production of toxins, proteolytic enzymes, and other factors that trigger inflammation, bone resorption, and immune system modulation.
- The bacterium can also play a role in systemic conditions, such as cardiovascular diseases, due to its ability to enter the bloodstream through periodontal lesions.
- Epidemiology:
- The prevalence of A. actinomycetemcomitans varies worldwide but is found in both healthy individuals and those with periodontal disease.
- Higher levels of this bacterium are usually detected in individuals with periodontal infections.
- Different serotypes (a-f) of A. actinomycetemcomitans have been identified, with some serotypes (like serotype b) being more strongly associated with periodontal disease.
- Diagnosis:
- Microbiological Culture: Using selective media to identify colonies.
- PCR and DNA Probes: For detecting specific genes or sequences unique to A. actinomycetemcomitans.
- Serological Methods: Identification based on serotype-specific antigens.
- Treatment:
- Mechanical Debridement: Removal of dental plaque and calculus from teeth.
- Antibiotics: Systemic antibiotics (like amoxicillin, metronidazole, or doxycycline) may be prescribed, especially in aggressive cases.
- Antimicrobial Mouth Rinses: Chlorhexidine or other antiseptic rinses can help reduce bacterial load.
- Host Modulation Therapy: Managing the host response to reduce tissue destruction.
- Prevention:
- Good oral hygiene practices, such as regular brushing, flossing, and professional dental cleanings, are critical in preventing the colonization and spread of A. actinomycetemcomitans.
Would you like more information on any specific aspect of A. actinomycetemcomitans?
Shedding Light on How Oral Bacteria Can Aggravate Rheumatoid Arthritis
Summary
Clinical studies have shown that periodontal disease with Aggregatibacter actinomycetemcomitans can exacerbate symptoms of rheumatoid arthritis (RA), but the molecular mechanisms behind this remain unclear. In a recent study, researchers from Japan conducted several experiments in a mouse model of arthritis to shed light on this topic. Their findings reveal the crucial role of macrophages and the protein caspase-11 in this context, hinting at promising therapeutic targets for RA and other periodontal infection-related diseases.
Periodontal disease, which affects the gums and tissues that surround the teeth, is one of the most prevalent dental conditions worldwide. Most often caused by the formation and accumulation of bacterial biofilm around the teeth, periodontal disease can ultimately lead to tooth loss if left unattended. Interestingly, the inflammatory effects of periodontal bacteria can go well beyond the mouth, leading to systemic effects. Over the past few decades, clinical studies have revealed that the periodontal pathogen Aggregatibacter actinomycetemcomitans (A. actinomycetemcomitans) is closely related to the onset and worsening of rheumatoid arthritis (RA), a serious autoimmune disease that affects joints. However, what goes down at the molecular level remains largely unexplored and unclear.
In a recent study published online on 15 August 2024, in International Journal of Oral Science, a research team from Tokyo Medical and Dental University (TMDU) in Japan sought to fill this knowledge gap through detailed mechanistic studies in an animal model.
First, the researchers conducted preliminary experiments to confirm whether A. actinomycetemcomitans infection influenced arthritis in mice. To this end, they used the collagen antibody-induced arthritis mouse model, which is a well-established experimental model that mimics several aspects of RA in humans. They found that infection with this specific bacterium led to increased limb swelling, cellular infiltration into the lining of the joints, and higher levels of the inflammatory cytokine interleukin-1β (IL-1β) within the limbs.
Notably, these symptoms of worsening RA could be suppressed by administering a chemical agent called clodronate that depletes macrophages—a type of immune cell. This demonstrated that macrophages were somehow involved in aggravating RA caused by A. actinomycetemcomitans infection.
Further investigation using macrophages derived from mouse bone marrow revealed that A. actinomycetemcomitans infection increased the production of IL-1β. In turn, this triggered the activation of a multiprotein complex known as the inflammasome, which plays a key role in initiating and modulating the body’s inflammatory response to infections.
The researchers added yet one more piece to this puzzle using caspase-11-deficient mice. In these animals, inflammasome activation due to A. actinomycetemcomitans was suppressed. Most importantly, caspase-11-deficient mice exhibited less deterioration of arthritis symptoms, hinting at the important role that caspase-11 plays in this context.
Our research findings provide new insights into the link between periodontal pathogenic bacteria and the exacerbation of arthritis through inflammasome activation, offering important information on the long-debated relationship between periodontal disease and systemic diseases.
Professor Toshihiko Suzuki, co-lead authors
Department of Bacterial Pathogenesis, Infection and Host Response,
Graduate School of Medical and Dental Sciences
Tokyo Medical and Dental University, Tokyo, Japan.
With any luck, these efforts will contribute to the development of novel therapeutic strategies to manage RA.
The findings of this research may pave the way for advances in clinical treatments for RA induced by infection with A. actinomycetemcomitans. Our suggestion to inhibit inflammasome activation could attenuate the expansion of inflammation to joints, resulting in a recovery from arthritis symptoms. Moreover, the outcome of our work could contribute to the development of treatment strategies for not only arthritis but also other systemic diseases, such as Alzheimer's disease, which is also related to periodontal pathogenic bacteria.
Dr. Tokuju Okano, co-lead author
Department of Bacterial Pathogenesis, Infection and Host Response,
Graduate School of Medical and Dental Sciences
Tokyo Medical and Dental University, Tokyo, Japan.
The article, “Caspase-11 mediated inflammasome activation in macrophages by systemic infection of A. actinomycetemcomitans exacerbates arthritis,” was published in International Journal of Oral Science at DOI: 10.1038/s41368-024-00315-x Caspase-11 drives interleukin-1β (IL-1β) secretion in paws and affects clinical severity of arthritis in mice infected with Aggregatibacter actinomycetemcomitans (A. actinomycetemcomitans)
Caspase-11 drives interleukin-1β (IL-1β) secretion in paws and affects clinical severity of arthritis in mice infected with Aggregatibacter actinomycetemcomitans (A. actinomycetemcomitans)
Researchers unveil mechanism by which periodontal bacterium A. actinomycetemcomitans exacerbated symptoms of rheumatoid arthritis (RA) conducted various experiments in a mouse model of rheumatoid arthritis (RA). They investigated the molecular-level effects of infection with the periodontal pathogen A. actinomycetemcomitans, and found that caspase-11 plays a key role in the exacerbation of RA, as mice deficient in this protein exhibited much milder symptoms.
AbstractIt's the same old paradox for creationists, repeated time and again in the world of pathogenic parasites such as these bacteria - is their pathogenicity the result of intelligent, therefore malevolent, design or is it the result of mindless, amoral natural processes in which no gods were involved and therefore no gods are culpable.
Clinical studies have shown that Aggregatibacter actinomycetemcomitans (A. actinomycetemcomitans) is associated with aggressive periodontitis and can potentially trigger or exacerbate rheumatoid arthritis (RA). However, the mechanism is poorly understood. Here, we show that systemic infection with A. actinomycetemcomitans triggers the progression of arthritis in mice anti-collagen antibody-induced arthritis (CAIA) model following IL-1β secretion and cell infiltration in paws in a manner that is dependent on caspase-11-mediated inflammasome activation in macrophages. The administration of polymyxin B (PMB), chloroquine, and anti-CD11b antibody suppressed inflammasome activation in macrophages and arthritis in mice, suggesting that the recognition of lipopolysaccharide (LPS) in the cytosol after bacterial degradation by lysosomes and invasion via CD11b are needed to trigger arthritis following inflammasome activation in macrophages. These data reveal that the inhibition of caspase-11-mediated inflammasome activation potentiates aggravation of RA induced by infection with A. actinomycetemcomitans. This work highlights how RA can be progressed by inflammasome activation as a result of periodontitis-associated bacterial infection and discusses the mechanism of inflammasome activation in response to infection with A. actinomycetemcomitans.
Introduction
Periodontitis is a chronic inflammatory disease caused by bacterial infection in the periodontium that leads to the secretion of numerous proinflammatory cytokines, such as IL-1β, TNFα, and IL-6, from gingival tissue or immune cells, resulting in the expansion of the inflammatory response systemically.1,2,3,4,5 Dental biofilms or plaques are required to induce periodontitis; however, recent studies based on independent metagenomic and mechanistic approaches have hypothesized that the pathogenesis of periodontitis includes polymicrobial synergy and the dysbiosis of periodontitis-associated bacteria.6 The dysbiosis of periodontal microbiota results a change in the relative abundance in health, leading to alterations in host-microbe crosstalk sufficient to mediate destructive inflammation and bone loss.7 Both the inflammatory response induced by periodontitis-associated bacteria and host-microbe crosstalk arising from polymicrobial synergy and dysbiosis are considered to play important roles in the pathogenesis of periodontitis. Periodontitis-associated bacteria are classified by their pathogenicity and properties into closely interrelated complexes. Especially, the highly pathogenic bacteria comprising the ‘red complex’ (Porphyromonas gingivalis, Treponema denticola, and Tannerella forsythia) are important in adult periodontitis, while Aggregatibacter actinomycetemcomitans can be a key factor in aggressive or juvenile periodontitis.1,8,9 Previous reports have shown that the spread of inflammation arising from periodontitis or periodontitis-associated bacterial infection can induce autoimmune diseases, including rheumatoid arthritis (RA), multiple sclerosis, infective endocarditis and ulcerative colitis; however, the critical molecular mechanisms are poorly understood.10,11,12,13
RA is a heterogenous chronic inflammatory autoimmune disease with the hallmarks of synovial inflammation and autoantibody production, leading to bone destruction. The activation of Toll-like receptors (TLRs) signaling and (NOD)-like receptor (NLR) signaling in a variety of cells, such as macrophages, dendritic cells, T cells and B cells, contributes to the chronic course of RA. The activation of these signaling pathways can be induced by microbial-associated molecular patterns (MAMPs), such as lipopolysaccharide (LPS) and peptidoglycan (PGN) expressed in microorganisms, or danger-associated molecular patterns (DAMPs),14 such as reactive oxygen species and ER stress generated in the cytosol. The activation of these signaling pathways also guides the secretion of proinflammatory cytokines and chemokines such as IL-1β, TNFα, IL-6, IL-8, and Type I interferons (IFNs), resulting in T-cell differentiation and activation, B cell activation, and autoantibody production. Autoantibodies against post-translationally modified protein antigens are necessary for RA to be triggered. A major example is anti-citrullinated protein antibodies (ACPAs). One report demonstrated that A. actinomycetemcomitans a common virulence factor, leukotoxin A (LtxA), produced by A. actinomycetemcomitans contributes to the generation of autoantigens that trigger RA, resulting in the progression of RA induced by cellular hypercitrullination in neutrophils; this previous report provided clinical data supporting a link between periodontal infection and RA.15 Other reports showed the similarity of periodontal or hematological characteristics such as cytokine production in blood between aggressive periodontitis and rheumatoid arthritis.16,17 Moreover, some clinical reports demonstrated that A. actinomycetemcomitans, P. gingivalis and T. forsythia were detected in peripheral blood from patients or healthy donor,18,19 suggesting the possibility that oral bacteria can spread from blood to localized places or oral to blood.
RA and periodontitis have much relation with inflammasome consists of a complex of caspase-1, nucleotide- binding oligomerization domain NLR family, and the adaptor protein apoptosis-associated speck-like protein containing a caspase recruitment domain (ASC) regulates caspase-1 cleavage and IL-1β secretion from cells by recognition of MAMPs or DAMPs.14,20,21,22,23,24,25,26 The inflammasome also controls pyroptosis, which is a cell death pathway executed by gasdermin D (GSDMD) in a manner that is dependent on cleaved caspase-1 and caspase-11.27,28 Recent studies have revealed that A. actinomycetemcomitans activates inflammasome NLR family pyrin domain containing 3 (NLRP3) or absent in melanoma 2 (AIM2) dependent manner in human macrophage by using small interference RNA analysis, and controls cell death through caspase-11 related pathway in murine macrophage by analysis of gene expression level, and murine alveolar bone loss by injected A. actinomycetemcomitans to gingival tissue was attenuated in NLRP3 or caspase-11-deficient mice, but relationships with pathogenesis for periodontitis or autoimmune diseases are elusive.29,30,31,32 In the present study, we demonstrated that LPS from A. actinomycetemcomitans activates the inflammasome in a caspase-11-dependent manner that is mediated by binding with CD11b and lysosomal degradation. Furthermore, we also discovered that inflammasome activation by infection with A. actinomycetemcomitans triggers progression of RA in a mice CAIA model.
Okano, T., Ashida, H., Komatsu, N. et al.
Caspase-11 mediated inflammasome activation in macrophages by systemic infection of A. actinomycetemcomitans exacerbates arthritis.
Int J Oral Sci 16, 54 (2024). https://doi.org/10.1038/s41368-024-00315-x
© 2023 Springer Nature Ltd.
Reprinted under the terms of s60 of the Copyright, Designs and Patents Act 1988.
Of course, we can rule out the childish biologically non-sensical excuse of 'sin' causing 'genetic entropy' and therefore 'devolution' from an initial perfect design since a perfect design should not be capable of deterioration and there is no credible mechanism by which detrimental mutations can spread though a species gene pool.
Despite decades of effort by the Deception Insitute to fool American politicians into thinking creationism is a science, not fundamentalist religion in a lab coat, all the daft notion of 'sin' affecting genetics does is show how it is exactly that. There is no force, substance or process called 'sin' known to science which can make chemistry and/or physics do anything. It is an entirely religious concept fundamental to the Abrahamic religions, Chrstianity, Islam and Judaism.
The Malevolent Designer: Why Nature's God is Not Good
Illustrated by Catherine Webber-Hounslow.
The Unintelligent Designer: Refuting The Intelligent Design Hoax





No comments :
Post a Comment
Obscene, threatening or obnoxious messages, preaching, abuse and spam will be removed, as will anything by known Internet trolls and stalkers, by known sock-puppet accounts and anything not connected with the post,
A claim made without evidence can be dismissed without evidence. Remember: your opinion is not an established fact unless corroborated.